Metformin Blocks ADMA to Switch Off mTOR and Switch on AMPK
That is the main reason it extends healthy life and delays chronic disease
WH Bestermann Jr MD, Jerry Reeves MD
Introduction
Metformin is the most prescribed medication for type 2 diabetes in the world. This much has been proven about the metformin-ADMA relationship. ADMA (asymmetric dimethylarginine) is increased in all cardiovascular risk factors. ADMA and metformin are so similar in structure that they neutralize each other. This opposition requires intact cells. It does not occur in isolated mitochondria. “The intriguing observation that metformin as a liposomal form was again able to exert its role when added directly to isolated mitochondria is in accordance with a membrane-mediated uptake and vesicular routing of metformin.”
“Metformin … inactivates mTOR through the AXIN/ LKB1-v-ATPase-Ragulator pathway” while also directly activating AMPK. Switched off mTOR is diffused in the cell. Switched on mTOR relocates to the lysosomal membrane. Arginine, ADMA, and metformin are all structural analogs. ADMA very likely switches on mTOR and switches off AMPK via the nutrient sensing complex v-ATPase-Ragulator. Metformin blocks that effect at an amino acid transporter-receptor (transceptor) in the lysosomal membrane. That is why metformin reduces the likelihood of heart attack and other diabetic complications so dramatically compared with achieving the same sugar level with other means. Metformin does not merely lower glucose and insulin resistance. It protects cells and organs by directly blocking the biochemistry that damages them. The rest of this article goes into these relationships in more detail.
Amino acids like arginine activate enzymes called Rags as part of the nutrient sensing mechanism that activates mTOR. ADMA is an arginine analog. “Metformin and phenformin inhibit RAG GTPase-mediated mTOR signaling.”
New research since the first article[1] on the ADMA-Metformin Hypothesis reveals more about the most likely mechanism of action of metformin. Metformin directly inhibits mTOR[2] (the mechanistic target of rapamycin) and activates AMPK[3] (AMP kinase). These master metabolic switches are critical for normal development. They coordinate growth and energy supply with available nutrients in juveniles. Global mTOR deletion is inconsistent with fetal survival.[4] mTOR becomes less active in healthy adults, only to be reactivated later in life by nutrient excess, reactive oxygen species, tobacco smoke, and age itself. The mTOR-AMPK axis is critical in aging[5] as well as chronic disease development[6] including cardiovascular diseases and cancer.[7] mTOR and AMPK are generally inversely related. Caloric restriction[8], exercise, metformin, ACE inhibitors, angiotensin receptor blockers[9], statins, and mineralocorticoid receptor antagonists[10] all directly or indirectly inhibit mTOR and activate AMPK. Combining lifestyle interventions, metformin, atorvastatin, an angiotensin converting enzyme inhibitor, and aspirin prolongs life and delays major cardiovascular events by eight years in patients with high-risk type 2 diabetes.[11]
Growth Factors and Adequate Nutrient Signaling Both Required for mTOR Activation
mTORC1 and mTORC2 are critical elements of the mTOR complex. Two parallel signaling pathways must be simultaneously active to switch on mTOR— the growth factor and nutrient sensing pathways.[12] The growth factor signaling pathway is well defined for several cardiometabolic elements. The AT1 receptor and mineralocorticoid receptor generate ROS which transactivate the epidermal growth factor receptor (EGFR). EGFR activates growth factor signaling to switch on mTOR. Angiotensin receptor blockers and mineralocorticoid receptor antagonists block this signaling. The small molecule metformin blocks a lysosomal transceptor (transporter/receptor) required for nutrient sensing and mTORC1 activation.
Brief ADMA Overview
ADMA (asymmetric dimethyarginine) is elevated in most cardiovascular risk factors and contributes to reactive oxygen species production and diminished NO activity. Some investigators believe it contributes to the pathology of cardiovascular disease[13]. ADMA levels correlate well with insulin resistance[14]. ADMA increases are a direct result of epigenetic activity. Most ADMA is formed when PRMT1 attaches methyl groups to arginine within proteins.[15] Arginine histone methylation opens DNA to allow transcription of genes involved with cell growth, proliferation, and apoptosis. ADMA is tightly correlated with normal growth in children.[16] ADMA levels go up acutely with certain foods leading to endothelial dysfunction within hours. [17]
ADMA is an Agonist antagonized by Metformin
ADMA, arginine, and metformin are structural analogs. (Figure 1) Metformin activity requires intact membranes and vesicular routing. Metformin engages a membranous site to reduce mitochondrial complex 1 activity. It requires intact cells for activity. It has no effect on isolated mitochondria but metformin in a liposomal form restored complex 1 inhibition. Metformin activity requires intact membranes and vesicular routing. ADMA at an identical concentration completely blocks metformin inhibition of complex 1.[18]
Metformin Directly Inhibits mTORC1 and Activates AMPK via Nutrient Sensing Machinery
Amino acids stimulate mTORC1 via a novel family of GTPases termed Rags. mTORC1 is one of two major subunits of mTOR. Metformin directly inhibits mTOR in a Rag GTPase-dependent manner[19] independent of TSC1/2 or AMPK. Rag GTPases mediate this response by inducing mTORC1 translocation from the cytoplasm to the lysosomal membrane. mTORC1 is diffused throughout the cytoplasm in amino acid depletion. Metformin treatment also causes mTORC1 diffusion even in the presence of amino acids.
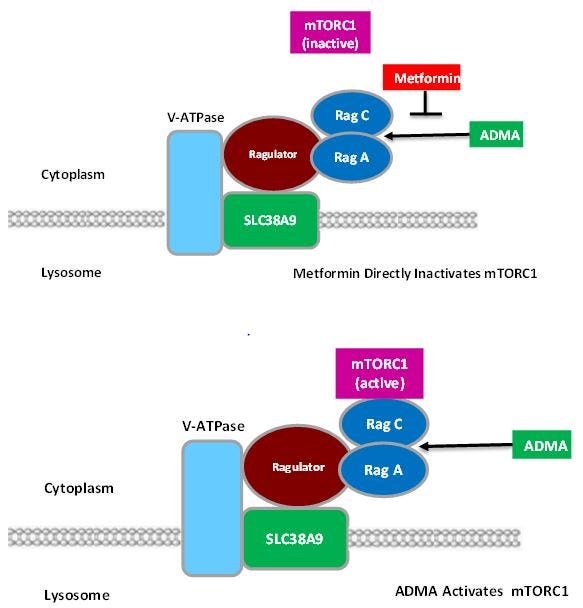
A lysosomal complex comprised of v-ATPase, Ragulator, SLC38A9, and Rag GTPase recruits mTORC1 to the lysosomal membrane in the presence of amino acids where it is activated by Rheb. The amino acid transporter SLC38A9 is an integral part of that complex . None of the other amino acid transporters tested, including SLC38A1, SLC38A2, SLC38A7, SLC36A1, SLC36A4 showed any affinity for the complex. It is the first member of the lysosomal amino acid sensing complex shown to physically engage amino acids. SLC38A9 silencing reduces amino acid mTORC1 activation. SLC38A9 is a component of the lysosomal amino acid complex that regulates mTORC1 activation. This transporter also has a receptor signaling function, so it is known as a transceptor.[20]
SLC38A9 binds arginine in the transmembrane region but it has a low affinity for arginine transport and likely does not control lysosomal arginine levels. It is not clear if the bound arginine comes from the lysosomal lumen or cytoplasm and both are likely. Arginine can enter lysosomes via a transporter or micropinocytosis. SLC38A9 arginine binding regulates mTOR activation and lysosomal efflux of several essential amino acid into the cytoplasm for protein synthesis. Arginine alone among the amino acids has this regulatory function.[21]
The N-terminal cytosolic region of SLC38A9 interacts strongly with the Rag GTPases and the amino acid sensing complex to activate mTOR signaling.[22] ADMA is an arginine analog[23] and it uses some of the same transporters as arginine[24]. SLC38A9 or another transceptor like it is the most likely site of ADMA mTOR activation.
It is likely that ADMA is the agonist that activates mTORC1 and metformin blocks ADMA activation at the SCL38A9 transceptor site. Metformin likewise directly activates AMPK by promoting formation of the v-ATPase-Ragulator-AXIN/LKB1-AMPK complex.[25] (Figure 2) That idea is further strengthened by the fact that ADMA added to adipocytes produces adipocyte hypertrophy and an increase in mTOR expression.[26] (Table 3)
Discussion
Additional research is needed to confirm and expand these relationships. At this point the ADMA-metformin relationship is still a hypothesis. It may not be entirely correct, but there is enough evidence to confirm ADMA and metformin oppose each other at the level of mTORC1 nutrient sensing. ADMA is a product of epigenetic processes that switch genes on acutely with certain foods and chronically with obesity. ADMA probably activates mTOR directly via a lysosomal transceptor and the Rag GTPase amino acid sensing system. That activation contributes to accelerated aging and earlier chronic disease onset. Metformin probably blocks ADMA and activates AMPK at the level of the lysosomal transceptor SLC38A9 which is part of the lysosomal mTORC1 activation complex on the lysosomal membrane. This makes metformin one of those highly specific small molecules like angiotensin II receptor blockers and mineralocorticoid receptor antagonists that block a specific receptor and reduce cardiorenal events more than they lower the target risk factor. These small molecules protect cells and organs. Metformin provides a tool to better understand nutrient sensing and mTORC1. The concept of the metabolic or cardiorenal syndrome understates the case. The mTOR-AMPK axis broadly impacts the molecular biology driving chronic disease and accelerated aging. Best practice treatments do not merely lower risk factors, they interfere with the molecular biology causing the damage that elevates the risk factor. Obese patients with hypertension or diabetes have elevated angiotensin II, aldosterone, and ADMA. AT1R blockade with very low dose angiotensin receptor blocker treatment does not lower the blood pressure, but it does interfere with signaling that causes complications.[27] Lisinopril and metformin also probably interfere with signaling at low dose without lowering the target risk factor. Intervening early with low doses of low-cost generic medication to block the signaling from these molecules is another form of precision medicine that can prolong healthy lives and delay the onset of chronic disease. Obese patients newly diagnosed with high blood pressure and/prediabetes already have arterial and/or pancreatic disease. Patients with high blood pressure should receive low dose losartan and spironolactone or eplerenone. Patients with prediabetes should add metformin. Early low doses should interfere with signaling and can be given much more safely early compared with later treatment when complications like CKD are already present. Something like this is already part of clinical practice. Recent guidelines recommend metformin for diabetes regardless of the glucose level, ACE inhibitor or ARB for chronic kidney disease regardless of the blood pressure, and statins for established vascular disease regardless of the LDL-cholesterol level. That all makes sense. It is just too little too late. The sooner molecular management begins, the longer we can all enjoy good health.
[1] Bestermann WH, The ADMA-Metformin hypothesis: Linking the cardiovascular consequences of the metabolic
syndrome and type 2 diabetes. Cardiorenal Med 2011;1(4):211-219 [pubmed 22135630]
[2] Kalender A, Selvaraj A, Kim SY Metformin, independent of AMPK, inhibits mTORC1 in a Rag GTPase-dependent
manner Cell Metab 2010;11(5):390-401 [pubmed 20444419]
[3] Zhang C-S, Li M, Ma T Metformin activates AMPK through the lysosomal pathway Cell Metab 2016;24:521-522 [pubmed 27732831]
[4] Gupta MB, Jansson T Novel roles of mechanistic target of rapamycin signaling in regulating fetal growth Biol
Reprod 2019;100(4):872-884 [pubmed 6698747]
[5] Longo VD, Antebi A, Barke A Interventions to slow aging in humans: Are we ready? Aging Cell 2015;14:497-510
[6] Stallone G, Infante B, Priciandaro C, Grandaliano G mTOR and aging: An old-fashioned dress Int J Mol Sci
2019;20:2774 [pubmed 6600378]
[7] Guanghong J, Aroor AR, Martinez-Lemus LA, Sowers JR Overnutrition, mTOR signaling, and cardiovascular
diseases Am J Physiol Regul Integr Comp Physiol 2014;307(10):R1198-R1206 [pubmed 4233289]
[8] De Cabo R, Mattson MP Effects of intermittent fasting on health aging and disease N Eng J Med 2019;381:2541-
2551 [pubmed 31940711]
[9] Ding G, Zhang A, Huang S, et al. ANG II induces c-Jun NH2-terminal kinase activation and proliferation of human
mesangial cells via redox-sensitive transactivation of the EGFR. Am J Physiol Renal Physiol. 2007;293(6):F1889‐
F1897 [pubmed 17881465]
[10] Huang S, Zhang A, Ding G, Chen R. Aldosterone-induced mesangial cell proliferation is mediated by EGF receptor
transactivation. Am J Physiol Renal Physiol. 2009;296(6):F1323‐F1333 [pubmed 19339632]
[11] Gaede P, Oellgaard J, Carstensen B et al Years of life gained by multifactorial intervention in patients with type 2
diabetes mellitus and microalbuminuria: 21 years follow-up on the Steno 2 randomized trial Diabetologia
2016:59(11)-2298-2307 [pubmed 27531506]
[12] Rebsamen M, Superti-Furga G A lysosomal amino acid transporter at the core of the amino acid-sensing
machinery that controls mTORC1 Autophagy 2016;12:1061-1062 [pubmed 5506099]
[13] Cooke JP Asymmetrical dimethyarginine: the uber marker? Circulation 2004;109:1813-1818 [pubmed 15096461]
[14] Stuhlinger MC, Abbasi F, Chu JW Relationship between insulin resistance and an endogenous nitric oxide
synthase inhibitor JAMA 2002;287:1420-1426 [pubmed 11903029]
[15] Blanc RS, Richard S Arginine methylation: The coming of age Mol Cell 2017;65:8-23 [pubmed 28061334]
[16] Gruber HJ, Mayer C, Meinitzer, et al. Asymmetric dimethyarginine (ADMA) is tightly correlated with growth in
juveniles without correlations to obesity related disorders Exp Clin Endocrinol Diabetes 2008;116(9):520-524
[17] Fard A, Tuck CH, Donis JA et al Acute elevations of plasma asymmetric dimethylarginine and impaired
endothelial function in response to a high-fat meal in patients with type 2 diabetes Arterioscl Throm Vasc Biol
2000;20:2039-2044 [pubmed 10978246]
[18] Detaille D, Guigas B, Leverve X, Wiernsperger N, Devos P Obligatory Role of Membrane Events in the regulatory
effect of Metformin on the Respiratory Chain Function Biochem Pharmacol 2002;63:1259-1272. [pubmed 11960602]
[19] Kalender A, Selvaraj A, Kim SY Metformin, independent of AMPK, inhibits mTORC1 in a Rag GTPase-dependent
manner Cell Metab 2010;11(5):390-401 [pubmed 20444419]
[20] Rebsamen M, Superti G SLC38A9: A lysosomal amino acid transporter at the core of the amino acid-sensing
machinery that controls mTORC1. Autophagy 2016;12:1061-1062 [pubmed 26431368]
[21] Wyant GA, Abu-Remaileh M, Wolfson RL et al mTORC1 activator is required to efflux essential amino acids from
lysosomes and use protein as a nutrient Cell 2017;171(3):642-654 [pubmed 5704964]
[22] Fan S-J, Goberdhan DCI PAT and SNATs: Amino acid sensors in disguise Front Pharmacol 2018;9:1-8
[23] Boger RH Asymmetric dimethylarginine (ADMA): A novel risk marker in cardiovascular medicine and beyond Ann
Med 2006;38(2):126-36
[24] Strobel J, Muller F, Zolk O Transport of asymmetric dimethyarginine (ADMA) by cationic amino acid transporter 2
(CAT2), organic cation transporter 2 (OCT2) and multidrug and toxin extrusion protein 1 (MATE1) Amino Acids
2013;45(4):989-1002.
[25] Zhang C-S, Li M, Ma T Metformin activates AMPK through the lysosomal pathway Cell Metab 2016;24:521-522
[26] Dowsett LB, Boruc O, Lee B, Leiper J Abstract 600:Asymmetric dimethylarginine is a novel regulator of mTOR
expression in adipocytes. Aterioscl Throm Vasc Biol 34 (Suppl 1) Abstract 600
[27] Saavedra JM Angiotensin II AT1 receptor blockers as treatments for inflammatory brain disorders Clin Sci (Lond)
2012;123(10):567-590





Agreed. Thank you for all you are trying to do! Merry Christmas and Happy New Year, Bill!
Thank you for this and every post!